The emergency linked to the contamination of infant formula by cereulide shows no signs of stopping. After the numerous recalls involving Nestlé in recent weeks, it is now also the turn of Aptamil and Mellin: the Ministry of Health has ordered the withdrawal of 76 batches, marketed by Danone Nutricia Spa.
The reason? The press release states that it is a “Adaptation to the new EFSA guidelines on cereulide limits“.
The point of the situation
For those who have not followed the story from the beginning, it is useful to make a quick summary. As of December 2025, dozens of countries around the world have initiated infant formula recalls due to the possible presence of cereulide, a toxin produced by the bacterium Bacillus cereus that can cause vomiting, diarrhea and dehydration in infants. Children under six months are particularly at risk.
The source of the contamination appears to be an oil rich in arachidonic acid (ARA) produced by a Chinese supplier and used by numerous companies in the formulation of infant milk. The fact that this ingredient was common to multiple manufacturers explains why the alert spread so quickly on a global scale, involving brands such as Nestlé and Lactalis.
The situation became even more dramatic in France, where two newborns who had consumed Guigoz brand milk (also owned by Nestlé) died. The judicial authorities have opened two investigations, although the causal link with the contaminated milk has not yet been established.
Following all this situation, on 2 February 2026, EFSA published the first official thresholds for cereulide in infant formula, setting the limit at 0.054 micrograms per liter for formula intended for the youngest infants.
These new analysis methodologies immediately produced concrete effects: Nestlé recalled a new batch of Nidina Optipro 1 the following day. Now it’s Aptamil and Mellin’s turn.
The new recalls
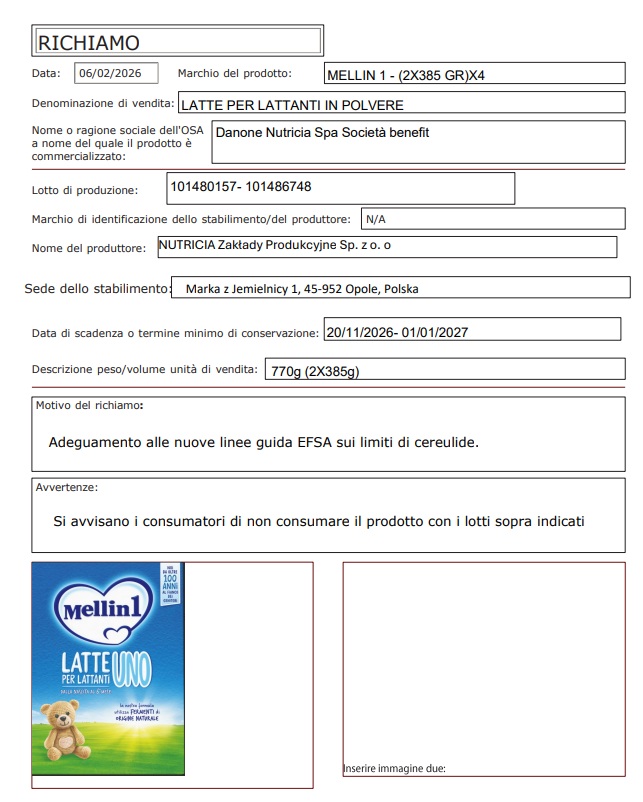
The Ministry of Health has issued 15 recall notices regarding Aptamil and Mellin products, all marketed by Danone Nutricia Spa and produced by Nutricia in three different factories in Poland, Ireland and the Netherlands. The reason is always the same: the possible presence of cereulide in an ingredient supplied by third parties.
Here are the products and lots affected:
Aptamil
Mellin
What parents should do
The advice of the health authorities is always the same: immediately check the batches of products at home and do not give the recalled packages to newborns. The recalled products can be returned to the point of sale where they were purchased.
Parents who notice concerning symptoms in children who have consumed one of the recalled batches – persistent vomiting, diarrhea, signs of dehydration – should contact their pediatrician immediately. If the symptoms are severe you need to go to the emergency room without waiting. As recalled by EFSA and ECDC, gastrointestinal disorders in the youngest newborns can degenerate rapidly, regardless of the underlying cause.
To stay updated on the recalled lots, it is always advisable to consult the Ministry of Health website, which publishes all recall notices in real time.
This new maxi recall confirms that the new thresholds set by EFSA on 2 February are having an immediate and concrete impact on the market. With more precise analytical parameters available, companies are now able to identify contamination that previously might have escaped detection, leading to more timely and widespread recalls.

