“Photo” to the cause of Parkinson’s: if not exactly like this, certainly today scientists led byUniversity of Cambridge (UK) have made a very important step forward in research on Parkinson’s disease. In fact, they managed to directly visualize and quantify the protein clusters that are believed to trigger the terrible neurology disease, which is currently the fastest growing in the world.
Parkinson’s disease and its symptoms
As our Istituto Superiore di Sanità (ISS) explains, Parkinson’s disease is a neurodegenerative disease characterized by muscular rigidity, which manifests itself with resistance to passive movements and tremor that occurs during the resting state, which in turn causes difficulty in starting and ending movements.
In turn, these symptoms induce balance disorders, awkward gait and hunched posture, sometimes combined with depression, also as an indirect consequence of one’s state of health, and slowness in speaking.
Recent research has also focused on this last symptom, demonstrating how changes in speech often occur before the characteristic symptoms appear, which is why some people with early-onset Parkinson’s may speak in a particular way and differently than usual.
A very debilitating symptom is also chronic pain, which affects a very high percentage of patients, between 70 and 80%. But this aspect is very often overlooked, as a study led by us has shown University of Verona.
How common is Parkinson’s disease
The Italian Society of Neurology reports that the pathology is the second neurodegenerative disorder in terms of frequency after the disease Alzheimer’sand which in industrialized countries has an incidence of approximately 12/100,000 people per year, with a prevalence of approximately 2 million affected individuals.
The disease is slightly more frequent in men than in women (60% vs 40%), and affects approximately 1% of the population over 60 years of age, reaching 4% among subjects over 85 years of age. But, although the prevalence progressively increases with age, cases in which the disease manifests itself before the age of 50, as well as before the age of 40 (juvenile Parkinson’s), are not rare.
By 2050, the number of people with Parkinson’s worldwide is expected to double, reaching 25 million.

What causes Parkinson’s disease
As with many neurodegenerative diseases, the causes of Parkinson’s are multiple and not fully understood. Some treatments have proven effective in controlling and slowing down the symptoms (unfortunately without being able to stop the progression), aimed above all at restoring dopamine levels, which, as now established, gradually decrease starting from the onset of the pathology.
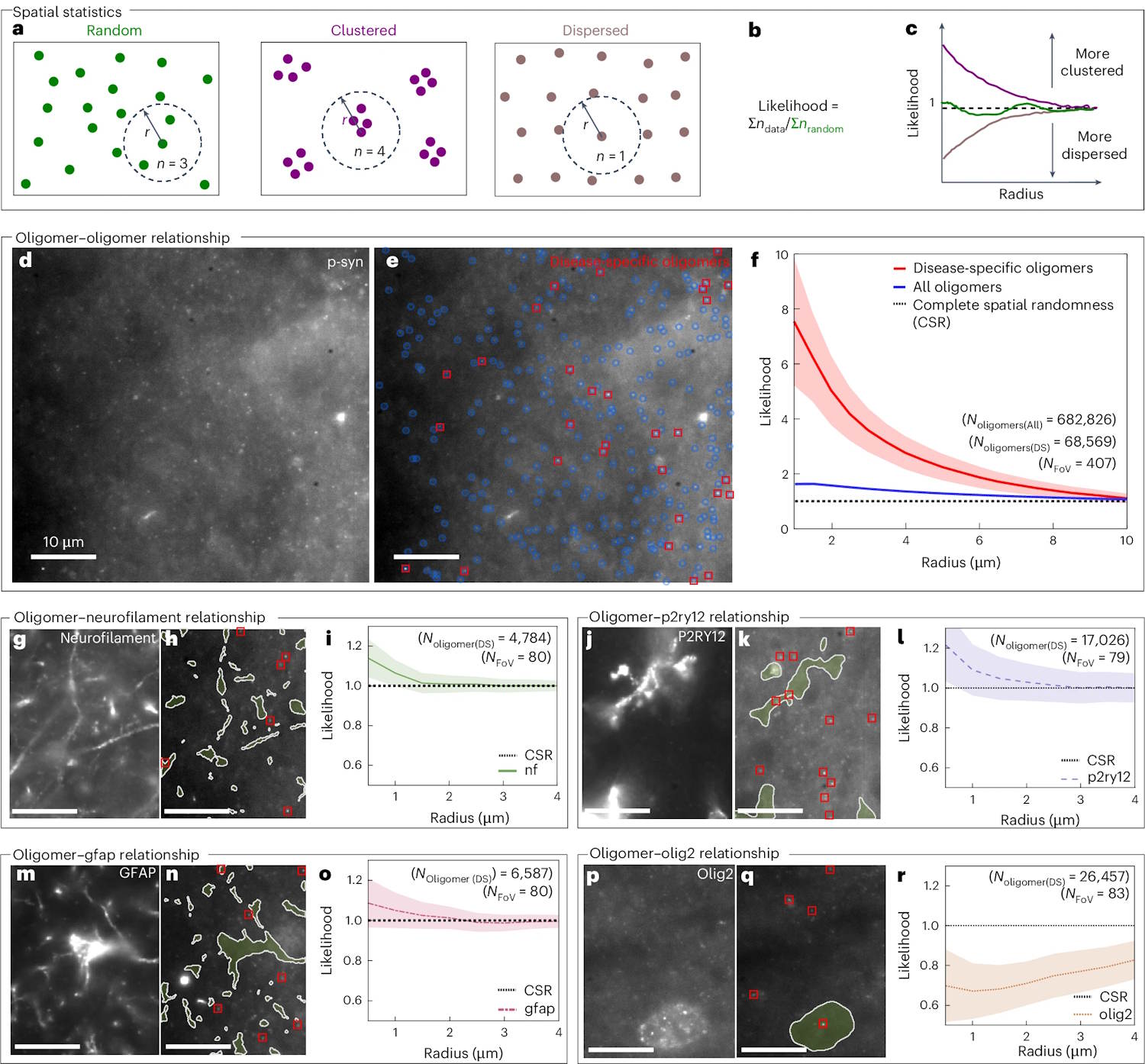
We also know that some tiny protein clusters, called alpha-synuclein oligomers, have long been considered likely responsible for the onset of Parkinson’s in the brain, but also for dementia with Lewyhowever they had until now escaped direct detection in human brain tissue.
A possible turning point
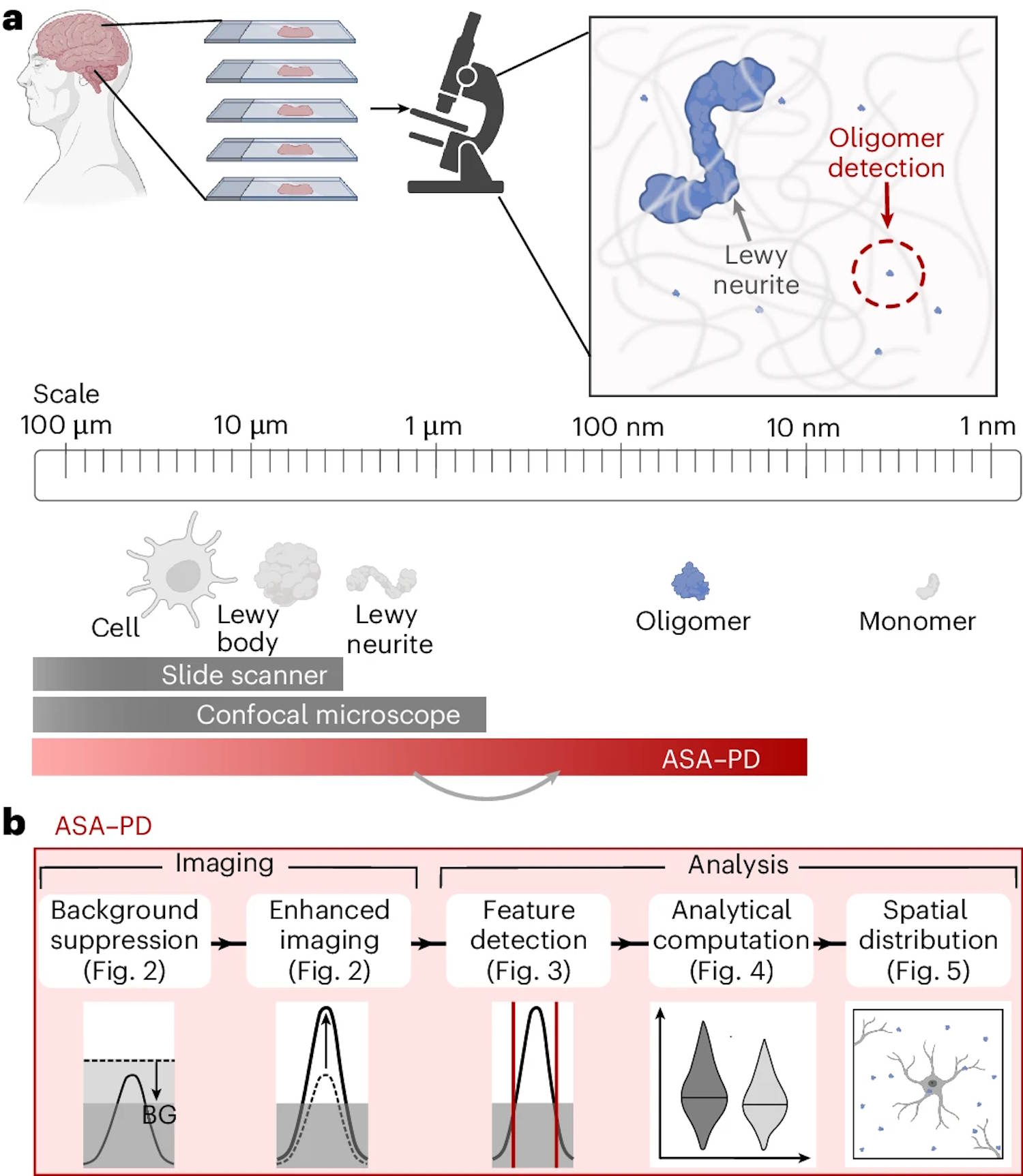
Now researchers have developed an imaging technique that allows them to see, count and compare oligomers in human brain tissue, a development that, according to one team member, is “like being able to see stars in broad daylight”.
These findings could help reveal how Parkinson’s spreads in the brain and support the development of diagnostic methods and potential treatments.
Lewy bodies are the hallmark of Parkinson’s, but they essentially indicate where the disease has gone, not where it is now – explains Steven Lee, who co-led the research – If we could look at Parkinson’s in its early stages, we could discover much more about how the disease develops in the brain and how we might be able to treat it
The developed technique, in particular, is called Advanced Sensing of Aggregates for Parkinson’s Disease (ASA-PD), and uses ultrasensitive fluorescence microscopy to detect and analyze millions of oligomers in brain tissue post-mortem.
Because the oligomers are very small, their signal is extremely weak, but ASA-PD maximizes the signal by reducing background noise, greatly increasing sensitivity to the point that it is possible to observe and study individual alpha-synuclein oligomers.
This is the first time we have been able to observe oligomers directly in human brain tissue at this scale: it’s like being able to see stars in broad daylight – reports Rebecca Andrews, first author of the work – All this opens new doors for Parkinson’s research
The team examined brain tissue samples post-mortem of people affected by Parkinson’s and compared them with those of healthy individuals of similar age, discovering that the oligomers are present in both the healthy brain and in the brain affected by Parkinson’s.
But above all, the main difference between the diseased and healthy brains is the size of the oligomers, which are larger, brighter and more numerous in the samples affected by Parkinson’s, which suggests a direct link with the progression of Parkinson’s.
The team also discovered a subclass of oligomers present only in Parkinson’s patients, which could be the first visible markers of the disease, potentially years before symptoms appear.
This method doesn’t just give us a snapshot – explains Lucien Weiss of the Polytechnique Montréal, who co-led the research – It offers a complete atlas of protein changes in the brain and similar technologies could be applied to other neurodegenerative diseases such as Alzheimer’s and Huntington’s disease
Oligomers have been the needle in the haystack, but now that we know where they are, they could help us target specific cell types in certain regions of the brain
The research was supported in part by Aligning Science Across Parkinson’s (ASAP), Michael J. Fox Foundation And Medical Research Council (MRC) and the work was published on Nature Biomedical Engineering.
Sources: University of Cambridge / Nature Biomedical Engineering

